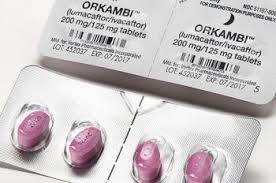
An event long waited in our community has finally happened. The targeted drug Orkambi that can be prescribed to cystic fibrosis patients with specific mutations has been approved in Russia.
Orkambi is an oral ivacaftor + lumacaftor combination. The Russian CF community has been waiting for it since 2015.
Of course, we understand that it’s still a long way to go until it is included in the List of Vital and Essential Drugs, and the clinical guidelines and standards are prepared. Still, it is our little victory and a reason to go on working!
Authorization technical data are available at the website of the Russian National Register of Medicinal Products.
To find out more on the significance of this drug authorization, read the following article on Takie Dela website — https://takiedela.ru/news/2020/12/17/orkambi/